
The carbon atoms present in our body were all once part of the carbon dioxide fraction of the atmosphere. Carbon is used to make explosives using principles of chemical science.Ībout 20% of the weight of living organisms is carbon.Īlmost all known compounds contain carbon. In the form of graphite, it is used in pencils. 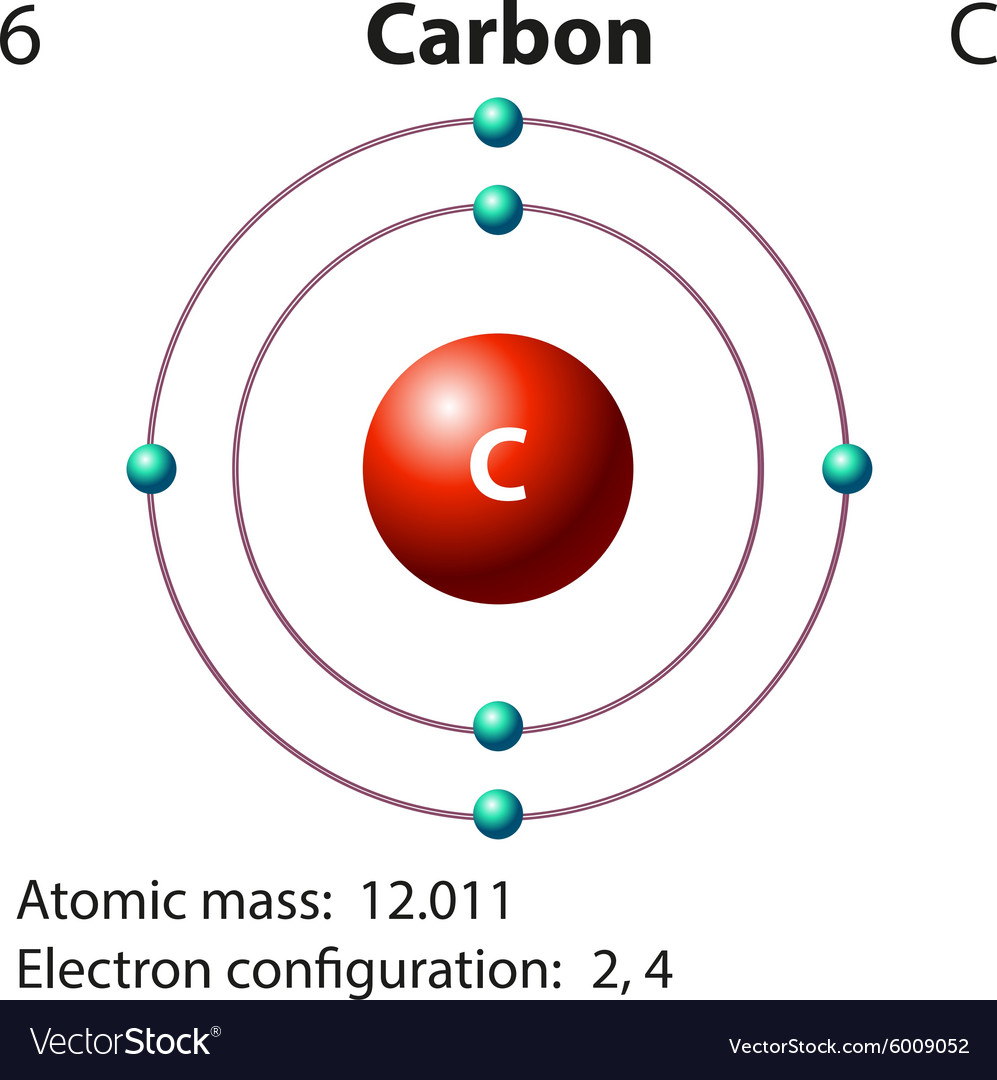
It is used as a gemstone (diamond) and for cutting and drilling. There are numerous applications of science involving carbon. Diamond is an excellent abrasive and it also has the highest thermal conductivity. It can bond with itself and numerous other elements forming over 9 million compounds.Ĭarbon is seen to be present in all living organisms and in science it is a very basic element in organic chemistry.Ĭarbon in its elemental form is present in nature as diamond, in its hardest form and as graphite which is the softest form. Here are some interesting facts about carbon for science students.Ĭarbon is a non-metal and its atomic number is six, its symbol is C. Let us consider the science about carbon, its composition. The atmosphere itself is constituted of about 0.03 % of carbon. Carbon was named by the renowned French Scientist Antoine Lavoisier and he carried out a variety of experiments to know about its nature.Ĭarbon is an essential and important part of a number of living things around us. During the ancient time, people, of course, did not realize that these substances were different forms of the same element. We know that a large number of living and non-living things in our environment are made of a common building block otherwise known as carbon.Ĭarbon has been known since ancient times in the form of soot, charcoal, graphite, and diamonds. Science is a study of living things and the constituents, processes in life around us.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
